Initial Treatment of Adults with HCV Infection Initial treatment of HCV infection includes patients with chronic hepatitis C who have not been previously treated with interferon, peginterferon, ribavirin, or any HCV directacting antiviral (DAA) agent, whether investigational, or US Food and Drug Administration (FDA) approvedDisoproxil or tenofovir alafenamide, represents the treatment of choice Pegylated interferonalfa treatment can also be considered in mild to moderate chronic hepatitis B patients Combination therapies are not generally recommended All patients should be monitored for risk of disease progression and HCCA Please refer to AASLD guidelines for recommended treatment regimens and durations 2 Identifying treatment candidates a Treatment is recommended for all patients with chronic HCV infection, except those with a short life expectancy who cannot be remediated by HCV therapy, liver transplantation, or another directed therapy b

Cldf
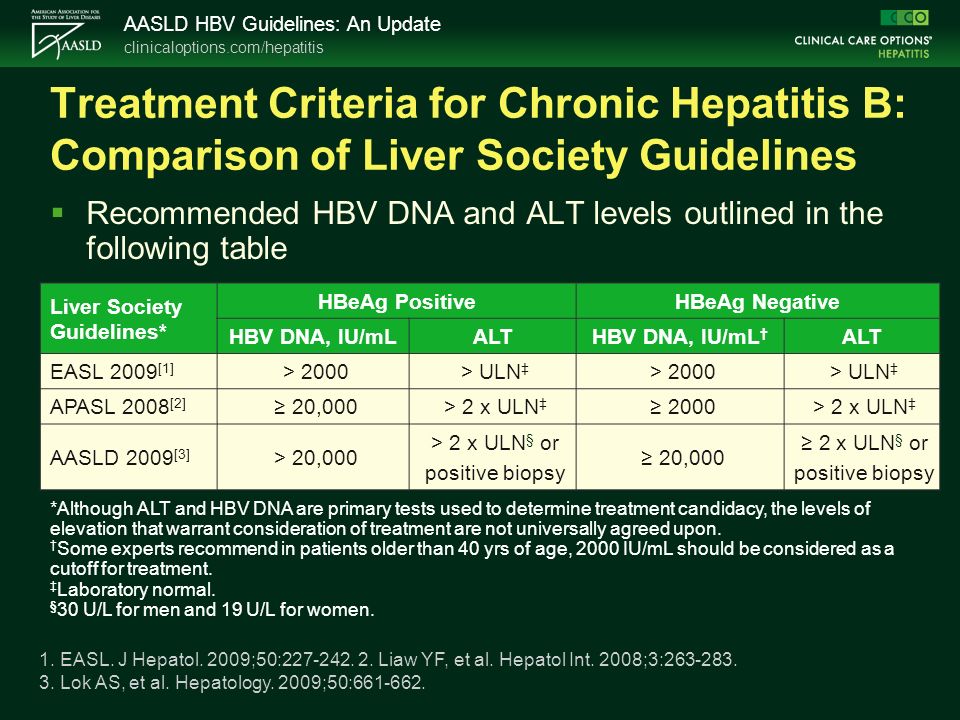
Aasld hep b treatment guidelines
Aasld hep b treatment guidelines-427 Acute hepatitis B All guidelines agree that patients with acute liver failure should receive antiviral therapy, and AASLD 18 guidance and APASL (APASL C2;AASLD Guideline16, for treatment of chronic Hepatitis B By Geeta Published On T Updated On 19 PM GMT People who test positive for the hepatitis B virus (HBsAg) for more than six months are



Hepatitis B Core Antibody Role In Clinical Practice In Springerlink
Hepbtalk 66 Comments The American Association for the Study of Liver Disease (AASLD), the organization that defines how doctors should treat hepatitis B and other liver ailments, unveiled new hepatitis B treatment guidelines this week at its annual conference in San Francisco The new guidelines are published hereNi YH, Chang MH, Wang KJ, et al Clinical relevance of hepatitis B virus genotype in children with chronic infection and hepatocellularAASLD Guidelines for Treatment of Chronic Hepatitis B Website View November 15 AASLD Guidelines for Treatment of Chronic Hepatitis B(link is external) Website View Three systematic reviews that were commissioned to support the guideline were published in January 16
Practice Guidelines Workshop New AASLD Guidelines A Summary of New Recommendations for Autoimmune Hepatitis, Women's Reproductive Health, Ascites, and KIC and Vascular Disorders of the Liver 0 – 330 pm AASLD/EASL Joint Symposium Steatohepatitis in Fatty Liver Disease In Europe and the US NIH Corner Parallel SessionsPatients on immunosuppressive agents are at risk for reactivation of HBV infection, and guidelines have been developed recommending routine pretreatment screening of patients for hepatitis B surface antigen (HBsAg) and antibodies to hepatitis B core antigen (antiH) Based on the serological profile (HBsAgpositive versus HBsAgnegative/antiHpositive) and the type, dose, and duration of immunosuppressive therapy, the risk of HBV reactivation during treatmentAnd (5) treatment of hepatitis B in special populations, including persons
The AASLDIDSA guidelines do not provide a recommendation regarding stopping or extending therapy in the setting of stable lowlevel viremia Determining Sustained Virologic Response The recommended testing to determine whether the patient has achieved an SVR is a quantitative HCV RNA level 12 weeks after completing therapyFor HBeAgnegative patients without cirrhosis, EASL guidelines suggest that stopping NA therapy may be considered in selected patients without cirrhosis who have undetectable serum HBV DNA for at least 3 years, provided close monitoring posttreatment is possible, whereas AASLD guidelines recommend indefinite treatment except for those whoGuidance for design and endpoints of clinical trials in chronic hepatitis B Report from the 19 EASLAASLD HBV Treatment Endpoints Conference ‡ J Hepatol Mar;72(3) doi /jjhep3




Hepatitis B Virus Whats New Easl Treatment Guidelines




Practice Guidelines sld
(3) specialized virological and serological tests;The 16 AASLD guidelines for the treatment of chronic hepatitis B as well as select recommendations from the 18 AASLD guidance update on the prevention, diagnosis, and treatment of chronic hepatitis B are outlined below and in the Guidelines sectionTerrault NA, Lok ASF, McMahon BJ, et al Update on prevention, diagnosis, and treatment of chronic hepatitis B AASLD 18 hepatitis B guidance Hepatology 18;



Frontiers Checkpoint Inhibitors And Therapeutic Vaccines For The Treatment Of Chronic Hbv Infection Immunology




Pdf Comparison Of Clinical Practice Guidelines For The Management Of Chronic Hepatitis B When To Start When To Change And When To Stop
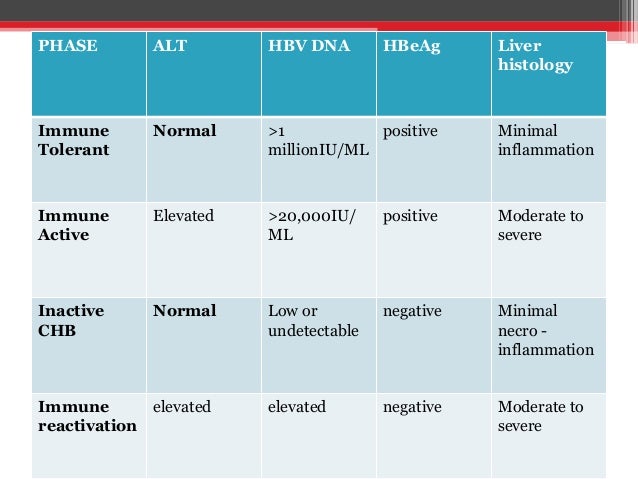
Hepatitis B virus (HBV) infection remains a global public health problem with changing epidemiology due to several factors including vaccination policies and migration This Clinical Practice Guideline presents updated recommendations for the optimal management of HBV infection Chronic HBV infectioThe 18 updated guidance on chronic hepatitis B (CHB) includes (1) updates on treatment since the 16 HBV guidelines (notably the use of tenofovir alafenamide) and guidance on (2) screening, counseling, and prevention;PRACTICEGUIDELINE AASLD Guidelines for Treatment of Chronic Hepatitis B Norah A Terrault,1 Natalie H Bzowej,2 KyongMi Chang,3 Jessica P Hwang,4 Maureen M Jonas,5 and M Hassan Murad6 See Editorial on Page 31 Objectives and Guiding Principles




sld Guidelines Hepatitis B 18 Ppt



Prevention Of Hbv Recurrence After Liver Transplant A Review
Diagnosis and Management of Autoimmune Hepatitis in Adults and Children 19Guideline Terrault NA, Lok ASF, McMahon BJ, et al Update on prevention, diagnosis, and treatment of chronic hepatitis B AASLD 18 hepatitis B guidance Hepatology 18 Apr 67 (4)Dr McMahon has served as coauthor of the AASLD Practice Guideline for Hepatitis B between 09 and 18 and is coAuthor of the AASLD Hepatitis B Guidance 19 He was also the cochair of the WHO Hepatitis Guideline published in 15




Practice Guidelines sld



Hepatitis B Cleveland Clinic
He is actively involved in clinical trials, teaching and having a busy clinical practice He has conducted more than 100 clinical trials in hepatology regarding chronic hepatitis C treatment, hepatitis B treatment and currently involved in many clinical trials with nonalcoholic fatty liver disease(3) specialized virological and serological tests;Members and Returning Customers Enter your Username and Password to login Here you can register for upcoming events, apply for membership, update your profile, manage your speaker invitations and disclosure information If you haven't accessed your account in the past six months, you will be asked to verify and/or update your contact information




Hepatitis B Core Antibody Role In Clinical Practice In Springerlink




Cldf
Updated AASLD hepatitis B guidance of 18 This AASLD 18 Hepatitis B Guidance is an update on AASLD 16 Practice Guidelines for Treatment of Chronic Hepatitis B1 and previous hepatitis B virus (HBV) guidelines from 09 The 18 updated guidance on chronic hepatitis B (CHB) includes (1) updates on treatment since the 16 HBV guidelines1 AASLD GUIDELINES FOR DIAGNOSIS &Now following APASL Guidelines have been published in Hepatology International !!!




Cost Effectiveness Analysis Of First Line Treatment For Chronic Hepatitis B In China Clinical Microbiology And Infection



sld Guidelines For Diagnosis Treatment Of Chronic Hepatitis B
AASLD guidelines for treatment of chronic hepatitis B AASLD guidelines for treatment of chronic hepatitis B AASLD guidelines for treatment of chronic hepatitis B Hepatology 16 Jan;63(1)261 doi /hep Epub 15 Nov 13 Authors Norah A Terrault 1On , the American Society of Clinical Oncology (ASCO) published provisional guidelines recommending that all people diagnosed with cancer be tested for hepatitis B before starting anticancer treatment According to the ASCO statement, up to 90% of people diagnosed with cancer have at least one risk factor for hepatitis B Cancer treatments can suppress theEASL evidence level II2, grade 1 recommendation) also include patients who have protracted severe acute hepatitis B (Table 5) WHO also support the use of antiviral therapy in fulminant or severe acute hepatitis




Understanding And Implementing The sld S Hbv Practice Guidelines Ppt Download



Early Detection Of Chronic Hepatitis B And Risk Factor Assessment In Turkish Migrants Middle Limburg Belgium
15 rowsAs for choice of antivirals, the majority of guidelines recommend theTreatmentNaive Patients Genotype 1b Without Cirrhosis a An 8week regimen can be considered in those with genotype 1b infection and mild fibrosis (see text for details) b Dosing is 3 coformulated tablets (glecaprevir 100 mg/pibrentasvir 40 mg) taken once daily Please refer to the prescribing informationTesting, Evaluation, and Monitoring of Hepatitis C HCV Testing and Linkage to Care When and in Whom to Initiate HCV Therapy Overview of Cost, Reimbursement, and CostEffectiveness Considerations for Hepatitis C Treatment Regimens Monitoring Patients Who Are Starting HCV Treatment, Are on Treatment, or Have Completed Therapy




Guidance For Design And Endpoints Of Clinical Trials In Chronic Hepatitis B Report From The 19 Easl sld Hbv Treatment Endpoints Conference Journal Of Hepatology



Management Of Hepatitis B Our Practice And How It Relates To The Guidelines
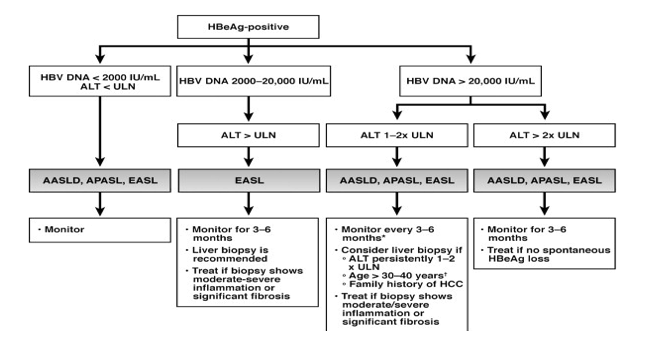
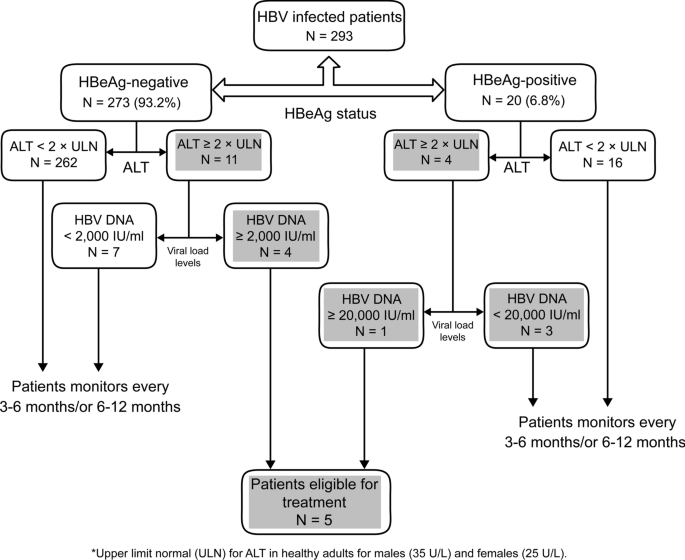
Guidance Statements for Treatment of Patients With Acute Symptomatic Hepatitis B Antiviral treatment is indicated for only those patients with acute hepatitis B who have acute liver failure or who have a protracted, severe course, as indicated by total bilirubin >3 mg/dL (or direct bilirubin >15 mg/dL), international normalized ratio >15, encephalopathy, or ascitesChronic hepatitis B HBsAg positive for more than six months, serum HBV DNA greater than ,000 IU per mL (lower values of 2,000 to ,000 IU per mL often occur with HBeAgnegative chronicTerrault NA, Bzowej NH, Chang KM, et al AASLD guidelines for treatment of chronic hepatitis B Hepatology 16;




Switching From Tenofovir Disoproxil Fumarate To Tenofovir Alafenamide In Virologically Suppressed Patients With Chronic Hepatitis B A Randomised Double Blind Phase 3 Multicentre Non Inferiority Study The Lancet Gastroenterology Hepatology



sld Guidelines For Diagnosis Treatment Of Chronic Hepatitis B
Guideline Terrault NA, Lok ASF, McMahon BJ, et al Update on prevention, diagnosis, and treatment of chronic hepatitis B AASLD 18 hepatitis B guidance Hepatology 18 Apr 67 (4)Sofosbuvir/velpatasvir was approved by the FDA for pediatric patients aged ≥6 years in March Given its pangenotypic activity, safety, and efficacy, sofosbuvir/velpatasvir is recommended as a first choice for HCV treatment in children and(4) monitoring of untreated patients;




Saslt Practice Guidelines For The Management Of Hepatitis B Virus Abaalkhail F Elsiesy H Alomair A Alghamdi My Alalwan A Almasri N Al Hamoudi W Saudi J Gastroenterol




Hepatitis B Virus Persistence And Reactivation The Bmj
(4) monitoring of untreated patients;JeanMichel Pawlotsky JeanMichel Pawlotsky, MD, PhD is professor of medicine at the University of ParisEst He is the director of the National Reference Center for Viral Hepatitis B, C and D and of the Department of Virology at the Henri Mondor University Hospital in Créteil, France, and director of research team "VirusesHepatologyCancers" at the Mondor Institute ofThe European Association for the Study of the Liver (EASL) hepatitis B clinical practice guidelines is the major hepatitis B guidance for Europe and this document was primarily written by gastroenterology and hepatology specialists The 17 EASL Hepatitis B Guidelines recommend initiating HBV treatment in the following situations




Easl 17 Clinical Practice Guidelines On The Management Of Hepatitis B Virus Infection Journal Of Hepatology




sld Guidelines
Guideline Terrault NA, Lok ASF, McMahon BJ, et al Update on prevention, diagnosis, and treatment of chronic hepatitis B AASLD 18 hepatitis B guidance Hepatology 18 Apr 67 (4)101–98) *This APASL Guideline canDr Tran has broad research interests in the areas of viral hepatitis B and C, liver disease in pregnancy and liver transplantation She is an internationally recognized expert and NIHfunded researcher in the field of chronic hepatitis B, and is active in patient and community advocacy on hepatitis B prevention and treatment




sld Recommendations For Treatment Of Chronic Hepatitis C Download Table




New Hepatitis B Treatment Guidelines Revealed At sld 15 Conference Hepatitis B Foundation
Current treatment guidelines of chronic hepatitis B the role of nucleos(t)ide analogues and peginterferon and treatment of chronic hepatitis B AASLD1Terrault NA, Lok ASF, McMahon BJ, et al Update on prevention, diagnosis, and treatment of chronic hepatitis B AASLD 18 hepatitis B guidance Hepatology , 18 2Terrault NA, Bzowej NH, Chang KM, et al AASLD guidelines for treatment of chronic hepatitis B Hepatology , 16Feb;71(2) doi /hep Authors Marc G Ghany 1 , Timothy R Morgan 2 , AASLDIDSA Hepatitis C Guidance Panel




Rebound Of Hbv Dna After Cessation Of Nucleos Tide Analogues In Chronic Hepatitis B Patients With Undetectable Covalently Closed Circular Dna Jhep Reports




Easl 17 Clinical Practice Guidelines On The Management Of Hepatitis B Virus Infection Journal Of Hepatology
Guideline Terrault NA, Lok ASF, McMahon BJ, et al Update on prevention, diagnosis, and treatment of chronic hepatitis B AASLD 18 hepatitis B guidance Hepatology 18 Apr 67 (4)And (5) treatment of hepatitis B inIn patients with chronic hepatitis B, estimates of the rates of HCV coinfection vary from 9% to 30% The primary concern with HBV/HCV coinfection is that it can lead to more severe liver disease and an increased risk for progression to liver cancer (HCC) Treatment of HBV/HCV coinfected patients can represent a challenge




Pdf South African Guideline For The Management Of Chronic Hepatitis B 13 Semantic Scholar



Prevalence Of Chronic Hepatitis B Phases In Eritrean Patients A Laboratory Based Cross Sectional Study Bmc Gastroenterology Full Text
Ahn J, Lee HM, Lim JK, et al Entecavir safety and effectiveness in a national cohort of treatmentnaïve chronic hepatitis B patients in the US the ENUMERATE study Aliment Pharmacol Ther 16; American Association for the Study of Liver Diseases website AASLD HBV Treatment Guidelines Published 18European Association for the Study of the Liver EASL 17 Clinical Practice Guidelines on the management of hepatitis B virus infection J Hepatol 17;The AASLD suggests indefinite antiviral therapy for adults with HBeAgnegative immuneactive chronic hepatitis B infection, unless there




Hepatitis C Guidance 19 Update American Association For The Study Of Liver Diseases Infectious Diseases Society Of America Recommendations For Testing Managing And Treating Hepatitis C Virus Infection Ghany



Hepatitis B Chapter 4 Yellow Book Travelers Health Cdc
TREATMENT OF CHRONIC HEPATITIS B 14 DR SREEJITH JR2 MEDICINE 2 • Official recommendations of AASLD on the treatment of chronic hepatitis B (CHB) virus (HBV) infection in adults and children • Multiple systematic reviews of literature were conducted 3APASL Guidelines for HBV "AsianPacific clinical practice guidelines on the management of hepatitis B a 15 update" (Hepatol Int 16;Media Contacts Nola Gruneisen, AASLD, 571‐292‐3068 Lauren Martin, IDSA, () HCVguidelinesorg — a website developed by the American Association for the Study of Liver Diseases and the Infectious Diseases Society of America to provide uptodate guidance on the management of hepatitis C — was recently revised to reflect important




Real World Single Center Experience On The Efficacy Of Stopping Long Term Nucleos T Ide Analog Therapy In Patients With Chronic Hepatitis B



Hepatitis B Treatments Now Available For Primary Care Bpacnz
Chronic hepatitis B (CHB) includes (1) updates on treatment since the 16 HBV guidelines (notably the use of tenofovir alafenamide) and guidance on (2) screening, counseling, and prevention;



The Asian Pacific Association For The Study Of The Liver Apasl




Table 3 From New Therapeutic Agents For Chronic Hepatitis B Semantic Scholar



Comparison Between Chronic Hepatitis B Patients With Untreated Immune Tolerant Phase Vs Those With Virological Response By Antivirals Scientific Reports




Con All Patients With Immune Tolerated Hepatitis B Virus Do Not Need To Be Treated Attar Clinical Liver Disease Wiley Online Library




Core Concepts Hepatitis B Coinfection Co Occurring Conditions National Hiv Curriculum



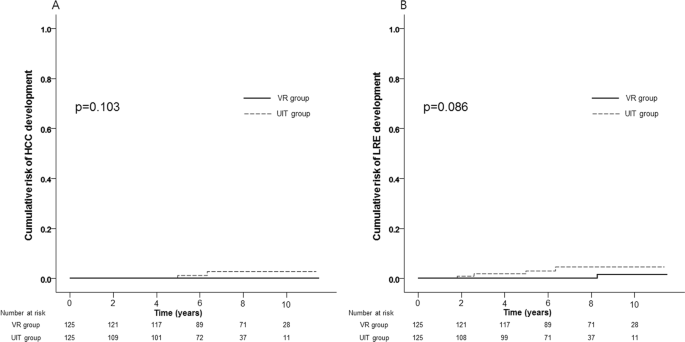
Management Of Concomitant Hepatocellular Carcinoma And Chronic Hepatitis C A Review




Clinical Guidelines Hepatitis B Foundation




Tenofovir Prophylaxis For Preventing Mother To Child Hepatitis B Virus Transmission In China A Cost Effectiveness Analysis International Journal Of Infectious Diseases




Practice Guidelines sld




sld Guidelines Hepatitis B Pregnancy




Practice Guidelines sld




Cldf
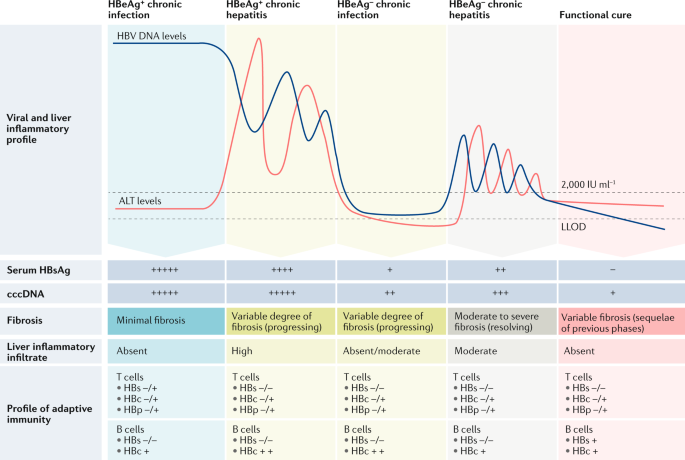



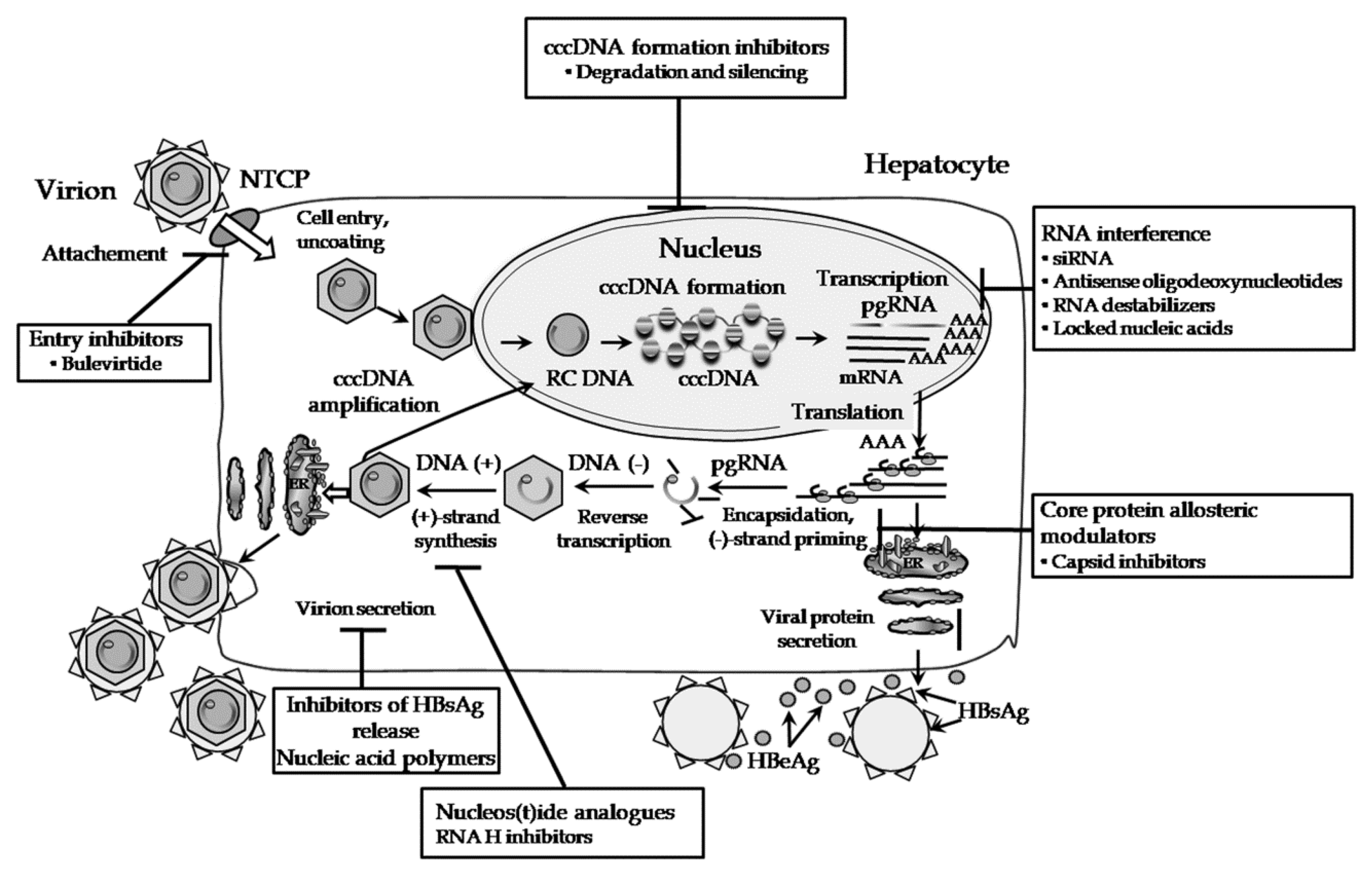
Therapeutic Strategies For Hepatitis B Virus Infection Towards A Cure Nature Reviews Drug Discovery




Guidance For Design And Endpoints Of Clinical Trials In Chronic Hepatitis B Report From The 19 Easl sld Hbv Treatment Endpoints Conference Sciencedirect




Clinicalthought Hbv Treatment Advances Clinicalthought sld Hepatitis Clinical Care Options




Hepatitis B Wikipedia
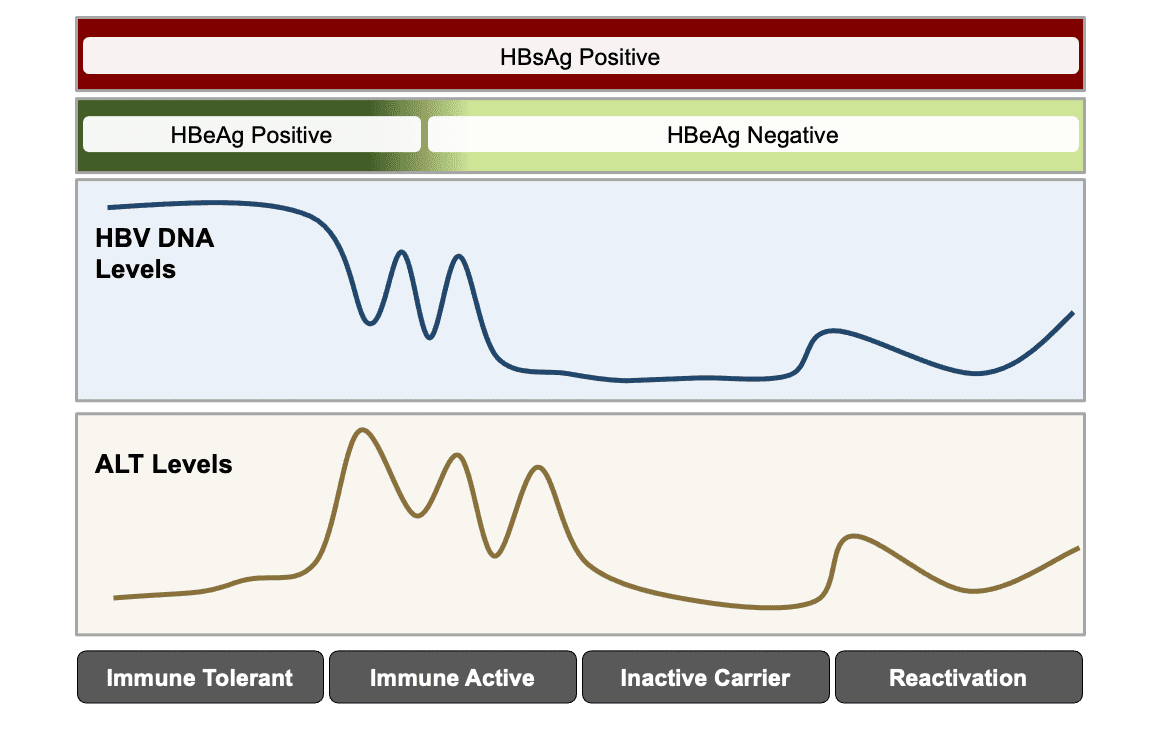



Core Concepts When To Initiate Hbv Treatment Treatment Of Hbv Hepatitis B Online




Experimental Drugs For The Treatment Of Hepatitis D Jep



South African Guideline For The Management Of Chronic Hepatitis B 13 Spearman South African Medical Journal



sld Guidelines Hepatitis B Reactivation



Jcm Free Full Text New Approaches To The Treatment Of Chronic Hepatitis B Html




Update On Prevention Diagnosis And Treatment Of Chronic Hepatitis B sld 18 Hepatitis B Guidance Terrault 18 Hepatology Wiley Online Library




Addressing The Global Burden Of Hepatitis B Virus While Developing Long Acting Injectables For The Prevention And Treatment Of Hiv The Lancet Hiv



Www Mdpi Com 1873 149x 27 1 2 Pdf



Q Tbn And9gctmrmzsbh1zirvbxaxhr5ctfegn7 Ai Lnode9rvfxy4qoj Rhh Usqp Cau




Hepatitis B Screening Prevention Diagnosis And Treatment American Family Physician




Hepatitis B And Pregnancy Virologic And Immunologic Characteristics Joshi Hepatology Communications Wiley Online Library




Hbv Treatment Hepatitis Clinical Care Options
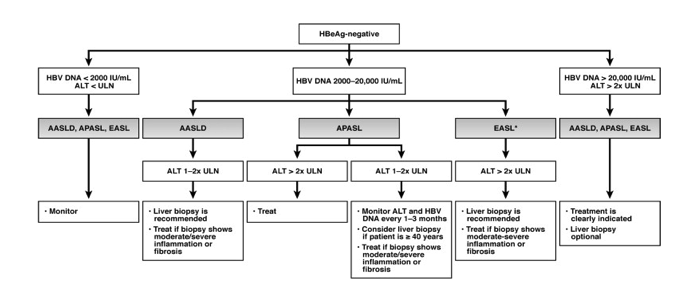



Management Of Hepatitis B Our Practice And How It Relates To The Guidelines




Comparison Of Clinical Practice Guidelines For The Management Of Chronic Hepatitis B When To Start When To Change And When To Stop
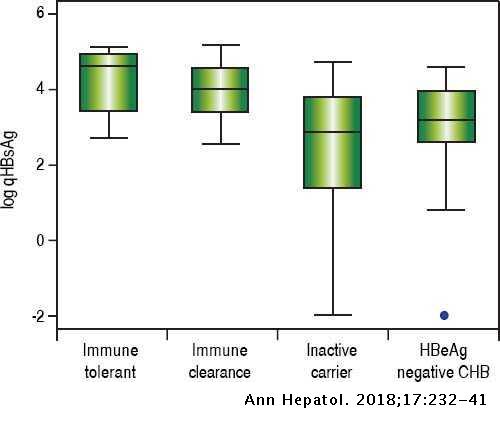



Long Term Follow Up And Quantitative Hepatitis B Surface Antigen Monitoring In North American Chronic Hbv Carriers Annals Of Hepatology
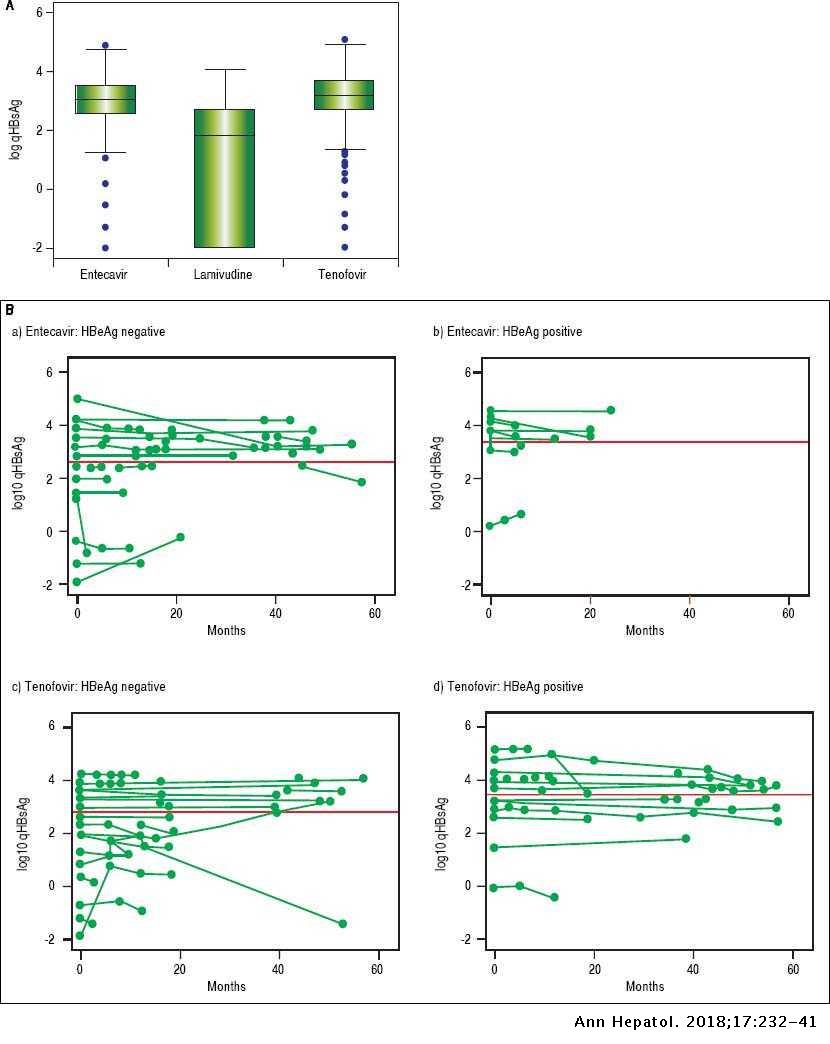



Long Term Follow Up And Quantitative Hepatitis B Surface Antigen Monitoring In North American Chronic Hbv Carriers Annals Of Hepatology
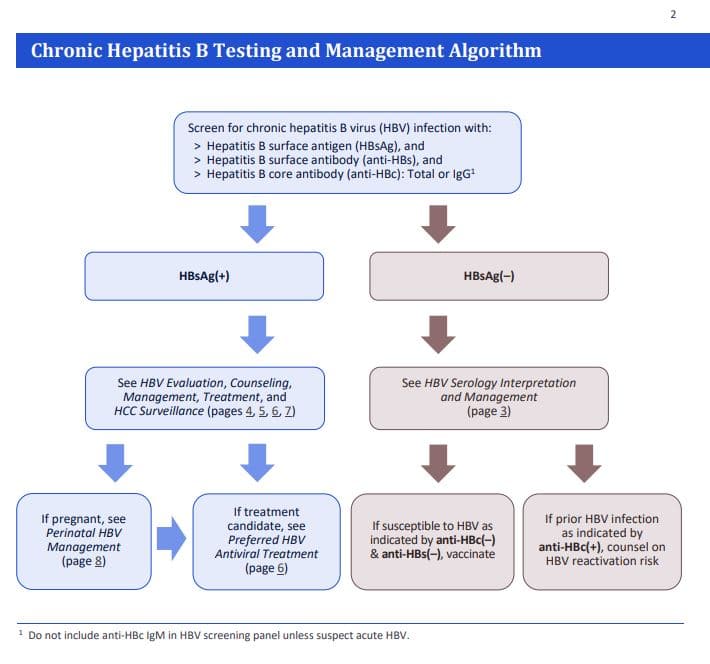



New Resource Guide To Hepatitis B Management For Primary Care Providers Hepatitis B Foundation




Pediatric Hepatitis B Treatment Komatsu Annals Of Translational Medicine




Kdoqi Us Commentary On The 18 Kdigo Clinical Practice Guideline For The Prevention Diagnosis Evaluation And Treatment Of Hepatitis C American Journal Of Kidney Diseases




Hepatitis B Secondary Prevention Wikidoc



1




Practice Guidelines sld




One World One Pandemic Many Guidelines Management Of Liver Diseases During Covid 19 Abstract Europe Pmc




Pdf Comparison Of Clinical Practice Guidelines For The Management Of Chronic Hepatitis B When To Start When To Change And When To Stop




sld Updates Chronic Hepatitis B Recommendations Practice Guidelines American Family Physician




Hepatitis B Related Outcomes Following Direct Acting Antiviral Therapy In Taiwanese Patients With Chronic Hbv Hcv Co Infection Journal Of Hepatology



Www sld Org Sites Default Files 19 06 sld 18 Hcc Guidance On Diagnosis 2c Staging And Management Hep 281 29 Pdf



Having Trouble Reading This Email View It In Your Browser November Contents News From The sld Liver Meeting Which People With Chronic Liver Disease Have Higher Covid 19 Risks Fatty Liver Greatly Increases Vulnerability To Covid 19 In




Hepatitis B Screening Prevention Diagnosis And Treatment American Family Physician




Hepatitis B 1 Chronic Hepatitis B Is A




Algorithm For The Management Of Hepatitis B E Antigen Hbeag Positive Download Scientific Diagram




Asian Consensus Recommendations On Optimizing The Diagnosis And Initiation Of Treatment Of Hepatitis B Virus Infection In Resource Limited Settings Gane Journal Of Viral Hepatitis Wiley Online Library




Hepatitis B Virus Advances In Prevention Diagnosis And Therapy Clinical Microbiology Reviews



Q Tbn And9gcqkchn Jjpqs9ezljmmawoolkrcgp Io4rht9opjhw Usqp Cau




Hepatitis B Virus From Diagnosis To Treatment



Infohep Some Hepatitis B Patients Can Safely Stop Antivirals



Q Tbn And9gcqxlq7ehbjyurwnjmgnm9ovgfge4l59duyvu9d1ztybh5lu9dy Usqp Cau



2



Early Detection Of Chronic Hepatitis B And Risk Factor Assessment In Turkish Migrants Middle Limburg Belgium
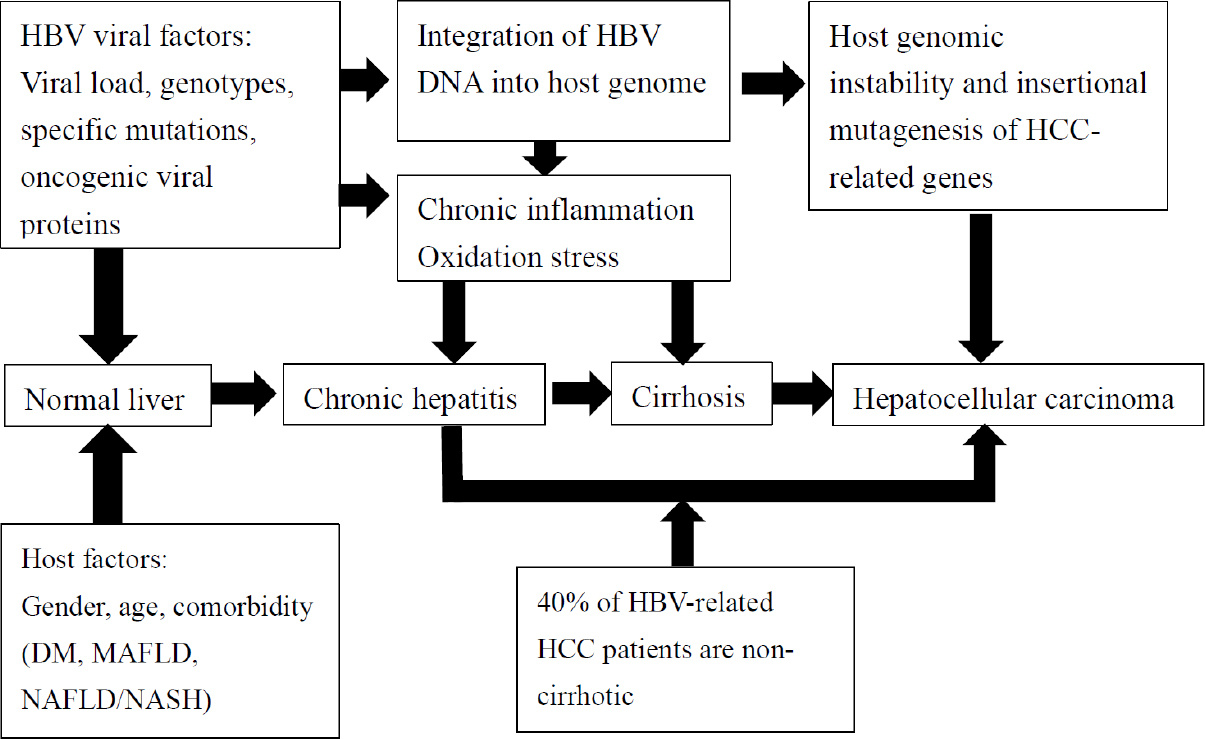



Prevention Of Hepatitis B Virus Related Hepatocellular Carcinoma




Easl 17 Clinical Practice Guidelines On The Management Of Hepatitis B Virus Infection Journal Of Hepatology




Hepatitis B Screening Prevention Diagnosis And Treatment American Family Physician




Guidelines For Patients With Immune Tolerant Chronic Hepatitis B Infection Download Scientific Diagram




Estimating The Proportion Of People With Chronic Hepatitis B Virus Infection Eligible For Hepatitis B Antiviral Treatment Worldwide A Systematic Review And Meta Analysis The Lancet Gastroenterology Hepatology




sld Posts Facebook
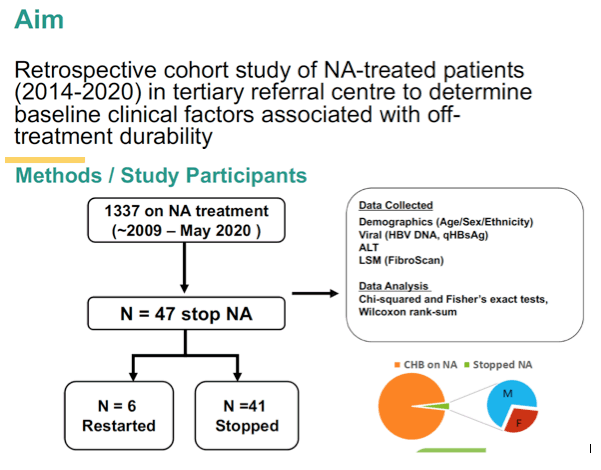



Real World Single Center Experience On The Efficacy Of Stopping Long Term Nucleos T Ide Analog Therapy In Patients With Chronic Hepatitis B
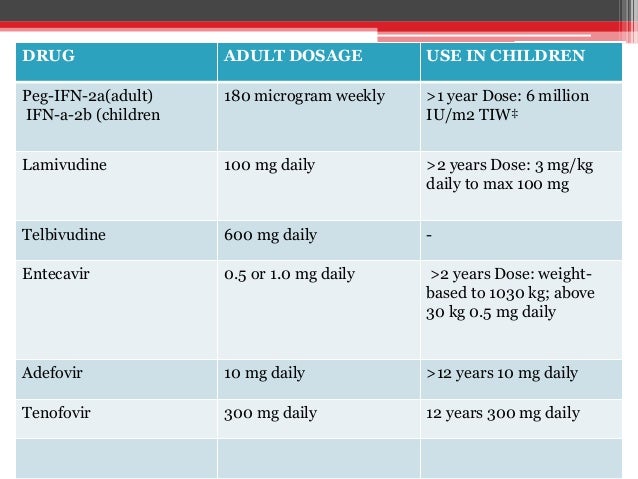



sld Guidelines For Diagnosis Treatment Of Chronic Hepatitis B




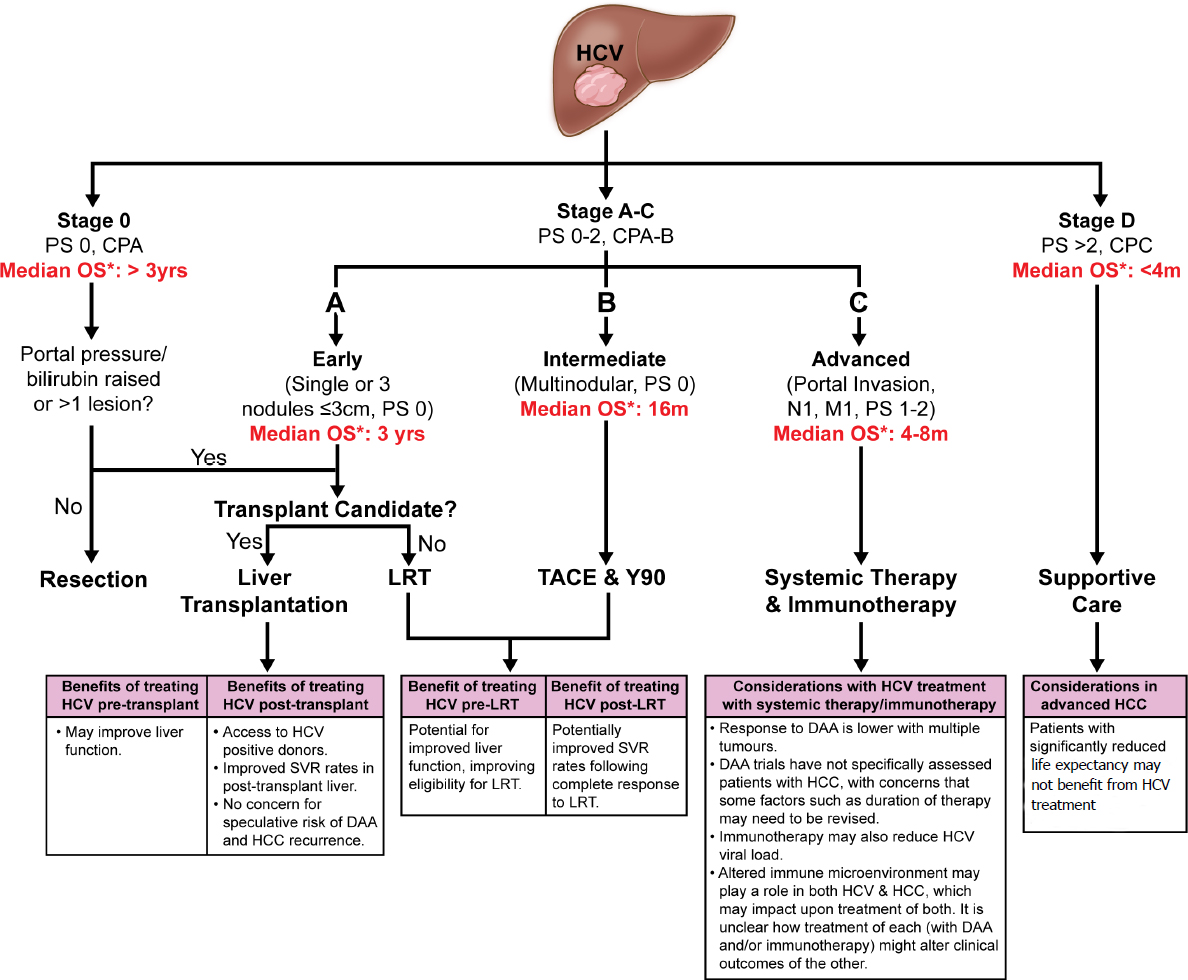
Comparison Of The Current International Guidelines On The Management Of Hcc Jhep Reports




Evolving Options For Hbv Therapy Navigating The New
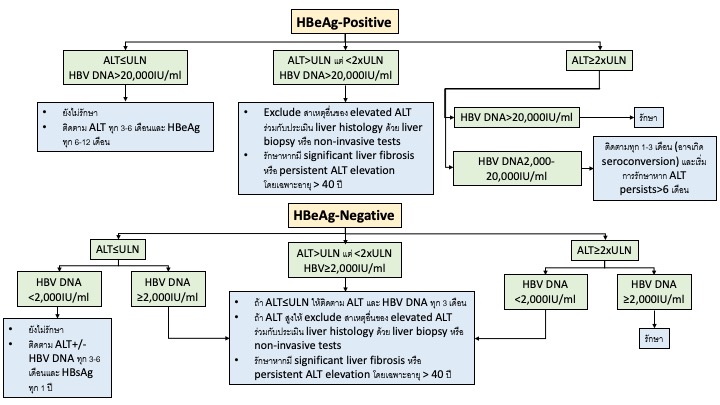



ไวร สต บอ กเสบบ Hepatitis B Infection สมาคมโรคต บแห งประเทศไทย
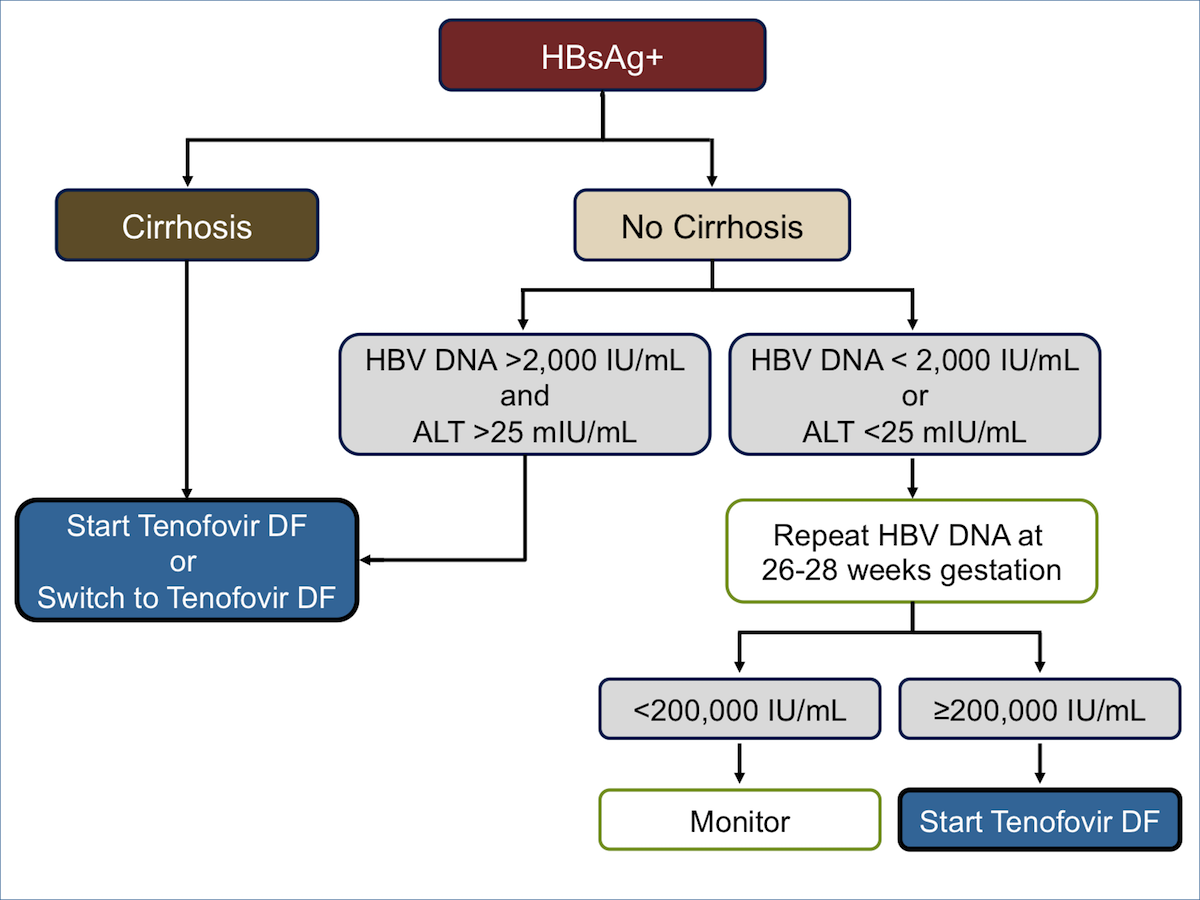



Core Concepts Preventing Hbv Perinatal Transmission Prevention Of Hbv Hepatitis B Online




Comparison Of Current Clinical Practice Guidelines For Chronic Download Scientific Diagram




Core Concepts Hepatitis B Coinfection Co Occurring Conditions National Hiv Curriculum




Understanding And Implementing The sld S Hbv Practice Guidelines Ppt Download




sld Cregg




Practice Guidelines sld



0 件のコメント:
コメントを投稿